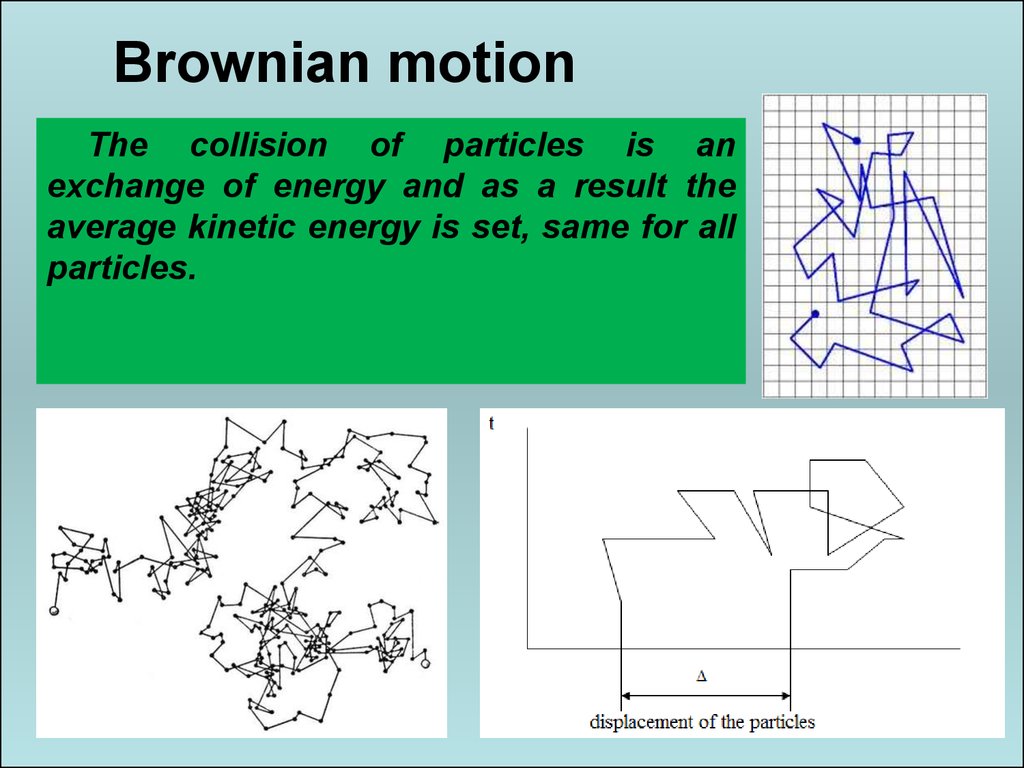
The particles usually less than 1 micrometer in size, and they suspended in a liquid or gas. The Brownian motion most easily observed in a suspension of small particles in a fluid. The Brownian motion named after the botanist Robert Brown, who first observed the phenomenon in 1828. The particles constantly buffeted by the surrounding fluid molecules, and this movement results in a chaotic jittering motion. The Brownian motion describes the random movement of particles suspended in a fluid. The tiny particles constantly buffeted by the molecules around them, and this motion causes them to move around chaotically. The Brownian Movement is the movement of tiny particles (such as atoms or molecules) in a liquid or gas, caused by the random motion of the molecules in the liquid or gas. 
What is the Brownian Movement in Chemistry? This movement is caused by the thermal energy of the particles. The particles are constantly moving around and bouncing off of each other. Motion of the pollen grain in water, movement of dust particles in a room, diffusion of pollutants in air are the Brownian motion examples.The Brownian Movement is the random movement of particles in a fluid.Brownian motion of the particles in a fluid depends on the size of the particle, density, viscosity and temperature of the fluid.Brownian motion can be calculated by parameter diffusion constant, which is given by.The unbalanced random force due to molecules of the fluid on the suspended particles causes it to move in an irregular manner.It was discovered by Robert Brown in 1827.The random or irregular motion of particles suspended in a fluid is called Brownian motion.Size of the particles: Brownian motion of the particles of bigger size is less to that of the particles of smaller size.Density of the fluid: Brownian motion of the particles increases with the decrease in density of the fluid and vice versa.Viscosity of the fluid: Brownian motion of the particles decreases with the increase in viscosity of the fluid in which particles are suspended and vice versa.Temperature of the fluid: Brownian motion of a particle increases with the increase in temperature and decreases with the decrease in temperature of the fluid.These are the factors which affects Brownian motion of the particles in a fluid: Therefore, temperature of the surrounding is given by Radius of the Brownian particle, r = 1.2 x 10 -10 m 
Given, Fluid viscosity, ɳ = 18 x 10 -6 Psĭiffusion constant, D = 0.5 x 10 -6 m 2 /s k B = 1.38 x 10 -23 JK -1, is Boltzmann constant and,īut, mobility of the particle of radius r in a fluid of coefficient of viscosity ɳ, is given byĪlso, Boltzmann constant, k B= \(\frac\).µ = mobility of the particles in the fluid,.It is also known as Brown noise or Red noise.īrownian motion of the particles suspended in a fluid can be calculated by a parameter known as the Diffusion constant ( D) and it is given by A low frequency sound is produced during the brownian motion of the particles, which is known as Brownian noise.Hence, the particles are subjected to an unbalanced force at each instant, causing them to move randomly.For the smaller particles, the number of molecules striking the various sides of the particle at any instant may not be equal.If the size of the suspended particles are sufficiently large then, an equal number of molecules strike the particles from each side at each instant.The suspended particles are extremely large in size as compared to the molecules of the fluids and these particles are continuously bombarded with the molecules of the fluids from all sides.Brownian motion can be defined as the irregular or uncontrolled movement of particles in fluid because of their constant collision with other fast moving molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
